
“Classic” alkali-silica reaction is a reaction between alkalis in cement and reactive silicate minerals in susceptible aggregates. The reaction creates a gel that absorbs water and expands, resulting in the characteristic three-armed cracks you can see in the photograph. This is the easiest way to diagnose ASR in the field.
In themselves, these cracks mainly affect serviceability. That’s because the worst cracking is near the surface of the concrete. However, these cracks may promote other modes of deterioration. For example, they could let in water, facilitating freeze-thaw damage. Or they might let in salts that promote corrosion of the reinforcement.
To get destructive alkali-silica-related expansions, you need three things:
- Susceptible aggregate
- A concrete pore solution with high pH
- Sufficient moisture.
It may be possible to obtain non-reactive aggregate, but it’s often cost prohibitive. As we use up nearby sources of good aggregates, we need to make better use of “good enough” aggregates.
Hydration of portland cement generates calcium hydroxide, which has a pH of about 12.5. Pozzolans in the concrete will react with the calcium hydroxide, but there will likely still be some left unreacted.
As for moisture, water is one of the essential ingredients in concrete. Some will react with the cementitious materials—that’s what hydration is—and some will evaporate. But even in dry environments, the relative humidity in mature concrete doesn’t usually get much lower than about 80%—high enough to promote alkali-silica reaction in susceptible aggregates.
Petrography identifies aggregates susceptible to alkali-silica reaction
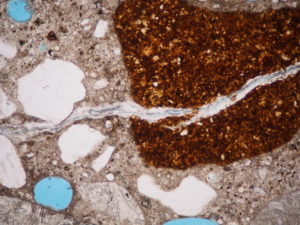
Not all siliceous minerals are equally susceptible to alkali-silica reaction. If the silica (SiO2) is in crystalline form, as quartz, it’s probably stable. However, if it’s glassy, as an opal or volcanic glass, it’s going to be highly reactive. Strained quartz and microcrystalline quartz are also reactive.
A petrographer can identify and quantify reactive forms of silica using ASTM C295, “Standard Guide for Petrographic Examination of Aggregates for Concrete.” The minerals to look out for are:
- Opal—more than 0.5% by mass
- Chert or chalcedony—more than 3.0% by mass
- Tridymite or christobalite—more than 1.0% by mass
- Optically strained or microcrystalline quartz—more than 5.0% by mass
- Natural volcanic glass—more than 3.0% by mass.
So how bad is it?
If the petrographic examination indicates a potentially reactive aggregate, you need to test it for expansion. ASTM C1260 and ASTM C1293 can identify those likely to cause deleterious expansions in concrete. In ASTM C1260, a 14-day expansion of 0.10% or less indicates an innocuous aggregate. In ASTM C1293, a 1-year expansion of 0.040% or less indicates an innocuous aggregate.
Even if you can’t wait a year for ASTM C1293 test results, you may be able to obtain them. State highway agencies build roads and bridges all over the state with local aggregates, so they have to know how they perform. And they’re usually willing to share the information. In addition, aggregate suppliers may already have test data on their materials. In either case, be sure the test pertains to the same seam of the quarry so it represents the same material.
Mitigating expansions due to alkali-silica reaction
Originally, engineers specified low-alkali cement—that is, cement with an equivalent alkali (Na2Oeq) content of 0.60%. However, there’s no guarantee that a low alkali content is low enough for a specific aggregate. And alkalis can be present in other concrete ingredients or penetrate the concrete from outside.
Fortunately, there are better ways to control expansions. Class F fly ash, some Class C fly ashes, slag cement, silica fume, and ground glass have all proved effective for this purpose. Whether in blended cements or added at the ready-mix plant, and whether in binary or ternary mixtures, they can all do the job. You just need to find out how much is enough. Strangely, the effects are not always proportional to the dosage. So you need to determine the optimum amount, not just the minimum.
Getting the performance you want
To determine the appropriate proportions of your materials with your aggregate, conduct a series of mortar-bar tests in accordance with 1567, “Standard Test Method for Determining the Potential Alkali-Silica Reactivity of Combinations of Cementitious Materials and Aggregate (Accelerated Mortar-Bar Method).” This is the same method as ASTM C1260, except you use your choice of supplementary cementitious material(s) instead of just portland cement. To save time, select a range of proportions and conduct the tests simultaneously. If the 14-day expansion is 0.10% or less, that combination should produce acceptable performance in service.
ASTM C1293 is also useful for evaluating combinations of supplementary cementitious materials in controlling expansions due to alkali-silica reaction. However, if you want to use it for this purpose, the exposure period is two years. While such a long evaluation period isn’t practical for most projects, test data from the aggregate supplier or the state DOT may provide guidance. ASTM C1778 provides a protocol for evaluating aggregates.
For projects in Minnesota, South Dakota, and Iowa, some aggregates are susceptible to alkali-silica-related popouts. These aggregates won’t be identified by ASTM C1260 or ASTM C1293. If your job site is in one of these states, contact us about identifying the aggregates and specifying appropriate procedures to avoid aggregate popouts.
Usually you want more from concrete than just control of alkali-silica reaction. Whether you need help writing the specification or you need to meet someone else’s specification, we can help you obtain all the results you need from your concrete.
